Genomic and functional fidelity of small cell lung cancer patient-derived xenografts.
Abstract
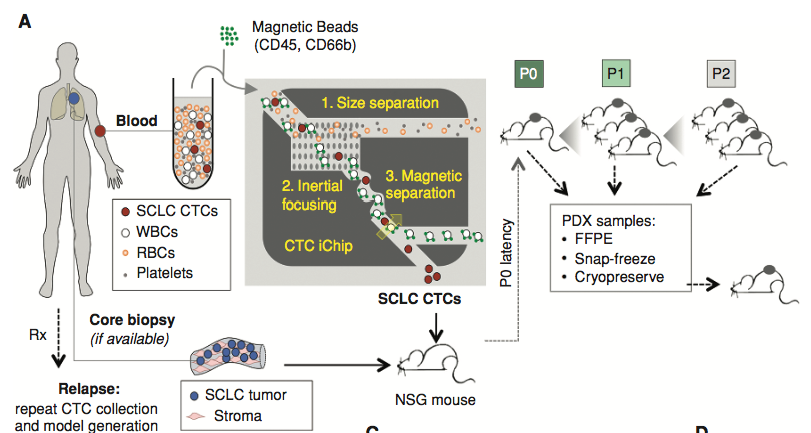
Small cell lung cancer (SCLC) patient-derived xenografts (PDXs) can be generated from biopsies or circulating tumor cells (CTCs), though scarcity of tissue and low efficiency of tumor growth have previously limited these approaches. Applying an established clinical-translational pipeline for tissue collection and an automated microfluidic platform for CTC-enrichment, we generated 17 biopsy-derived PDXs and 17 CTC-derived PDXs in a two-year timeframe, at 89% and 38% efficiency, respectively. Whole exome sequencing showed that somatic alterations are stably maintained between patient tumors and PDXs. Early-passage PDXs maintain the genomic and transcriptional profiles of the founder PDX. In vivo treatment with etoposide and cisplatin (EP) in 30 PDX models demonstrated greater sensitivity in PDXs from EP naïve patients, and resistance to EP corresponded to increased expression of a MYC gene signature. Finally, serial CTC-derived PDXs generated from an individual patient at multiple time points accurately recapitulated the evolving drug sensitivities of that patient’s disease. Collectively, this work highlights the translational potential of this strategy.
Contribution: RNAseq processing and analysis